NA • 322393

| Product name | B-hHER2 B16-F10 |
|---|---|
| Catalog number | 322393 |
| Strain name | NA |
| Strain background | C57BL/6 |
| NCBI gene ID | (Human) |
| Chromosome | 11 |
| Aliases | Neu; HER2; HER-2; c-neu; Erbb-2; c-erbB2; l11Jus8; mKIAA3023 |
| Tissue | Skin |
| Disease | Melanoma |
Origin: The B16-F10 cell line is derived from C57BL/6 murine skin cells. The cell line is a commonly used murine model for melanoma.
Background Information: The human epidermal growth factor receptor 2 (HER2), known as erbB-2, or proto-oncogene Neu, is a receptor tyrosine-protein kinase encoded by the ERBB2 (HER2) gene on chromosome 17q12. ErbB2 does not bind ligand, yet appears to be the major signaling partner for other ERBB receptors by forming heteromeric complexes with ErbB1, ErbB3, or ErbB4. Upon ligand-induced dimerization, the receptors auto phosphorylate on specific tyrosine residues in their cytoplasmic tails. These residues provide docking sites for phosphotyrosine binding, cytoplasmic signaling molecules that activate numerous intracellular signaling pathways. HER2 overexpresses in a broad number of cancer types, such as bladder, breast, cervical, cholangio, colorectal, endometrial, esophageal, gastric, head and neck, liver, lung, ovarian, and salivary gland cancers. Especially, amplification and overexpression of HER2 occurs in 25% to 30% of the instances of human breast cancer and are associated with a poor prognosis. Several HER2-targeting therapies such as trastuzumab, pertuzumab, T-DM1, DS8201 and RC48 have been approved worldwide for patients with HER2-positive tumors.
Gene targeting strategy: The exogenous CAG promoter and human HER2 coding sequence were inserted to replace part of murine exon 2 and all of exons 3-7. The insertion disrupts the endogenous murine Her2 gene, resulting in a non-functional transcript.
Tumorigenicity: Confirmed in B-hHER2 mice.
Application: The B-hHER2 B16-F10 tumor models can be used for preclinical evalsuation of ADC and monoclonal antibody drugs targeting human HER2.
Notes:
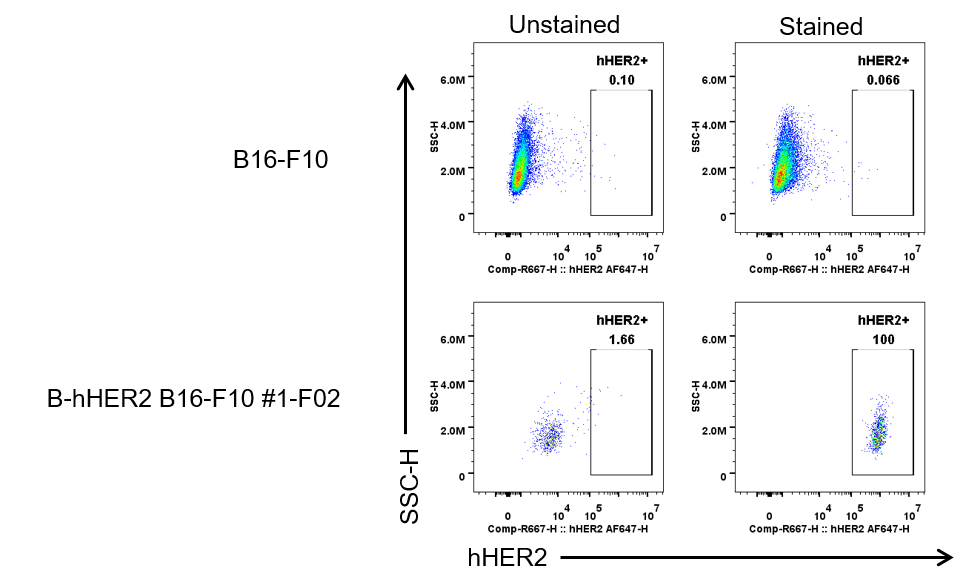
HER2 expression analysis in B-hHER2 B16-F10 #1-F02 by flow cytometry. Single cell suspensions from wild-type B16-F10 and B-hHER2 B16-F10 #1-F02 cultures were stained with human HER2 Antibody (Pertuzumab, in house). Human HER2 was detected on the surface of B-hHER2 B16-F10 #1-F02 cells, but not on the surface of wild-type B16-F10 cells. The clones of B-hHER2 B16-F10 #1-F02 cell was used for in vivo tumor growth assays.
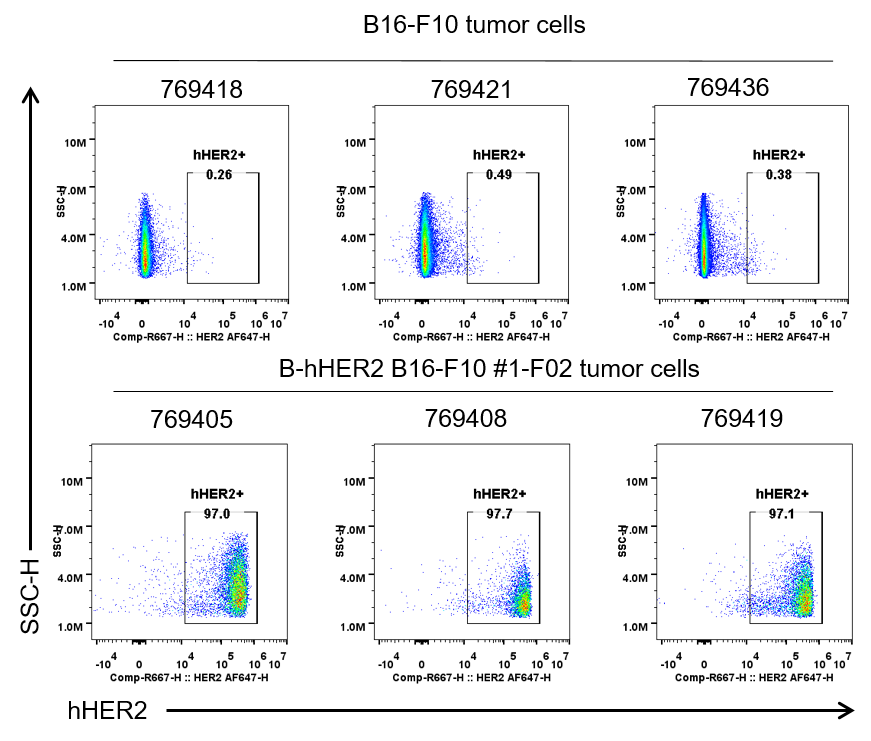
HER2 expression were evalsuated on B-hHER2 B16-F10 tumor cells by flow cytometry. B-hHER2 B16-F10 cells were subcutaneously transplanted into B-hHER2 mice (n=8). Upon conclusion of the experiment, tumor cells were harvested and assessed for human HER2 expression by flow cytometry (Pertuzumab, in house). As shown, human HER2 was highly expressed on the surface of tumor cells. Therefore, B-hHER2 B16-F10 cells can be used for in vivo efficacy studies evalsuating novel HER2 therapeutics.

Subcutaneous tumor growth of B-hHER2 B16-F10. B-hHER2 B16-F10 (2×105) and wild-type B16-F10 cells (2×105) were subcutaneously implanted into B-hHER2 mice (female, 8-week-old, n=8). Tumor volume and body weight were measured thrice a week. (A) Average tumor volume. (B) Body weight. Volume was expressed in mm3 using the formula: V=0.5 × long diameter × short diameter2. Results indicated that B-hHER2 B16-F10 cells were able to establish tumors in vivo and can be used for efficacy studies. Values are expressed as mean ± SEM.

Antitumor activity of anti-human HER2 antibody (DS8201) in B-hHER2 mice. (A) Anti-human HER2 antibody inhibited B-hHER2 B16-F10 tumor growth in B-hHER2 mice. Murine melanoma B-hHER2 B16-F10 cells were subcutaneously implanted into homozygous B-hHER2 mice (female, 9-week-old, n=6). Mice were grouped when tumor volume reached approximately 150 mm3, at which time they were intravenously injected with anti-human HER2 ADC DS8201 (purchased from Daiichi Sankyo) indicated in panel. (B) Body weight changes during treatment. As shown in panel A, anti-human HER2 ADC DS8201 was efficacious in controlling tumor growth in B-hHER2 mice in a dose-dependent manner, demonstrating that B-hHER2 B16-F10 cells provide a powerful preclinical model for in vivo evalsuation of anti-human HER2 ADC. Values are expressed as mean ± SEM.